Trending...
- Red5 Taps PubNub to Power the Next Era of Real-Time Interactive Streaming
- Colorado: Governor Polis Celebrates Agriculture Day, Signs New Laws Strengthening Agricultural Support
- New Book Reveals The Science Of Predictions
NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP) $NRXP: FDA Alignment, 70,000-Patient Real-World Data, and a Debt-Free Balance Sheet Position NRXP for Transformational 2026 Catalysts
MIAMI - ColoradoDesk -- In a pivotal development that could redefine the treatment landscape for suicidal depression, NRx Pharmaceuticals (N A S D A Q: NRXP) has emerged from a high-level, in-person Type C meeting with the U.S. Food and Drug Administration with what management believes is a clear path toward New Drug Application (NDA) submission for NRX-100 (preservative-free ketamine)—supported by both prior clinical trials and an unprecedented real-world dataset exceeding 65,000 patients.
For investors, the implications are substantial: regulatory clarity, expanding indications, strategic partnerships, a strengthened balance sheet, and a global ketamine market estimated at $750 million annually—with no FDA-approved drug currently indicated specifically for suicidal ideation.
FDA Type C Meeting Signals Regulatory Momentum
NRXP's meeting was attended by leadership from the FDA Division of Psychiatry Products and the Center for Drug Evaluation and Research (CDER)—a strong signal of institutional engagement at the highest levels.
Key takeaways:
This broader indication meaningfully expands the addressable patient population and commercial potential.
NRX-100 has already received Fast Track Designation for the treatment of acute suicidality in depression and bipolar depression.
70,000 Patients: Real-World Data at Scale
NRXP licensed data from more than 70,000 U.S. patients treated with IV ketamine or nasal S-ketamine for depression and suicidal ideation.
Preliminary analysis of a 20,000-patient subset demonstrated:
Notably, there is currently no FDA-approved medication specifically indicated for suicidal ideation, with Electroconvulsive Therapy (ECT) remaining the primary intervention.
More on Colorado Desk
If FDA alignment continues, NRX-100 could become the first drug positioned specifically for this indication under Accelerated Approval.
KETAFREE™: First Preservative-Free Ketamine
NRXP has applied for the proprietary name KETAFREE™, designed to be the first preservative-free ketamine formulation submitted for approval in this setting.
Given existing clinical familiarity with ketamine, the transition from generic compounded products to an FDA-reviewed, preservative-free branded formulation presents a compelling commercialization thesis.
Dual Strategy: Drug Development + Clinic Network Expansion
In parallel, NRXP announced a joint offering with neurocare Group AG to create a nationwide network of integrated neuroplastic care clinics targeting:
The model integrates:
Recent publications demonstrate:
NRXP plans to integrate neurocare clinics with HOPE Therapeutics and leverage an installed base of 400+ Apollo® TMS machines nationwide, creating a scalable service revenue channel alongside drug commercialization.
NRX-101: Breakthrough Therapy with Expanded Market Potential
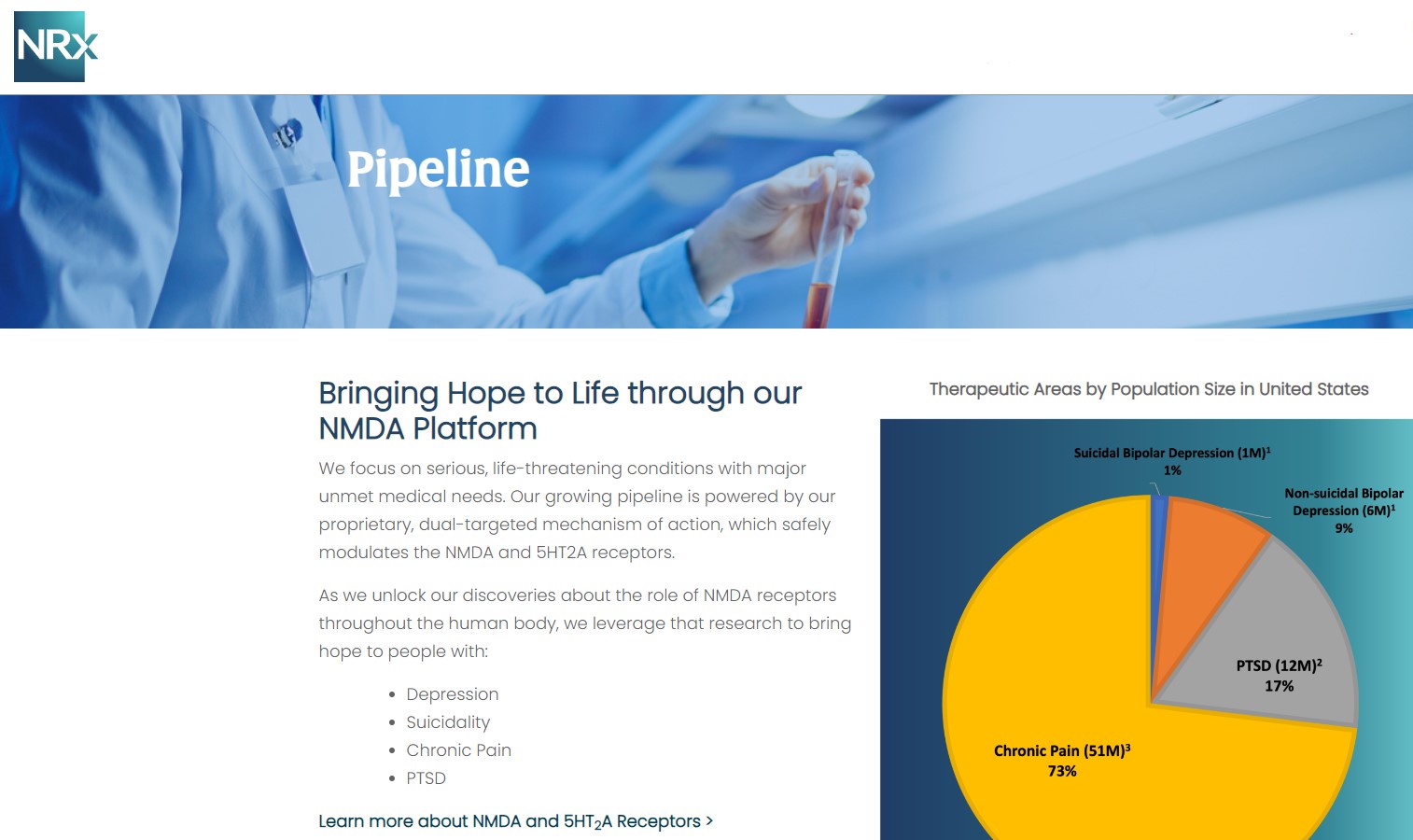
Beyond NRX-100, NRXP is advancing NRX-101, an FDA-designated Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain.
Key attributes:
With projections suggesting 1 million Americans annually may receive TMS by 2030, NRX-101 could capture a significant share of a rapidly expanding neurostimulation market.
NRXP has partnered with Alvogen Pharmaceuticals for development and marketing of NRX-101 in suicidal bipolar depression.
Debt-Free Balance Sheet: Strategic Reset for Growth
More on Colorado Desk
In December, NRXP eliminated its remaining $5.4 million in balance sheet debt via equity conversion—with no warrants or toxic provisions.
This clean capital structure positions the company for:
Analyst Coverage: $34 Price Target
Investment firm D. Boral has issued a Buy rating with a $34 price target, citing regulatory progress, expanded indications, and commercial optionality.
For a company operating at the intersection of FDA reform, real-world evidence utilization, and neuroplastic treatment innovation, this represents a high-conviction thesis on both regulatory and market catalysts.
The Macro Opportunity
According to CDC data, more than 13 million Americans seriously consider suicide each year.
Yet there is no FDA-approved drug indicated specifically for suicidal ideation.
NRXP is positioning itself to:
This is not a single-asset story—it is a platform strategy targeting one of the largest and most urgent unmet needs in psychiatry.
Investment Thesis Snapshot
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Media Contact
Company Name: NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Contact Person: Matthew Duffy, Chief Business Officer
Company Website: https://www.nrxpharma.com/
Email: mduffy@nrxpharma.com
Phone: (484) 254-6134
Home Country: United States
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
For investors, the implications are substantial: regulatory clarity, expanding indications, strategic partnerships, a strengthened balance sheet, and a global ketamine market estimated at $750 million annually—with no FDA-approved drug currently indicated specifically for suicidal ideation.
FDA Type C Meeting Signals Regulatory Momentum
NRXP's meeting was attended by leadership from the FDA Division of Psychiatry Products and the Center for Drug Evaluation and Research (CDER)—a strong signal of institutional engagement at the highest levels.
Key takeaways:
- Path to NDA based on existing adequate and well-controlled trial data
- Submission supported by 65,000+ patient Real-World Evidence (RWE) dataset
- No additional nonclinical studies required
- No bridging studies needed for preservative-free formulation
- Opportunity to seek broader indication: Treatment-Resistant Depression (TRD) with suicidality—not just acute suicidality
This broader indication meaningfully expands the addressable patient population and commercial potential.
NRX-100 has already received Fast Track Designation for the treatment of acute suicidality in depression and bipolar depression.
70,000 Patients: Real-World Data at Scale
NRXP licensed data from more than 70,000 U.S. patients treated with IV ketamine or nasal S-ketamine for depression and suicidal ideation.
Preliminary analysis of a 20,000-patient subset demonstrated:
- Rapid resolution of depression
- Rapid reduction in suicidal ideation
- Clinical outcomes consistent with prior randomized trials
- Favorable comparison to currently approved therapies
Notably, there is currently no FDA-approved medication specifically indicated for suicidal ideation, with Electroconvulsive Therapy (ECT) remaining the primary intervention.
More on Colorado Desk
- Senior Legacy Transition Program Introduced by Denver Metro Realtor with RE/MAX Professionals
- PandaGuarantee Launches Rent Guarantor Service in New York City
- The $112M Marketing Lesson Joe Whyte Learned: Why 'More Traffic' Is the Biggest Lie in Digital Marketing
- Colorado: Protecting Public Safety: Governor Polis Signs Social Media Law to Support Law Enforcement and Enhance Safety
- Daniel Kaufman Expands Kaufman & Company Real Estate Platform With New Acquisitions, AI-Driven Industrial Development and Nationwide Growth Initiative
If FDA alignment continues, NRX-100 could become the first drug positioned specifically for this indication under Accelerated Approval.
KETAFREE™: First Preservative-Free Ketamine
NRXP has applied for the proprietary name KETAFREE™, designed to be the first preservative-free ketamine formulation submitted for approval in this setting.
Given existing clinical familiarity with ketamine, the transition from generic compounded products to an FDA-reviewed, preservative-free branded formulation presents a compelling commercialization thesis.
Dual Strategy: Drug Development + Clinic Network Expansion
In parallel, NRXP announced a joint offering with neurocare Group AG to create a nationwide network of integrated neuroplastic care clinics targeting:
- Depression
- PTSD
- Bipolar Depression
- Autism
- Traumatic Brain Injury
The model integrates:
- Transcranial Magnetic Stimulation (TMS)
- Ketamine and neuroplastic drugs
- Hyperbaric oxygen therapy
- Supportive psychotherapy
Recent publications demonstrate:
- 87% clinical response
- 72% remission
in treatment-resistant depression when combining TMS with neuroplastic therapies.
NRXP plans to integrate neurocare clinics with HOPE Therapeutics and leverage an installed base of 400+ Apollo® TMS machines nationwide, creating a scalable service revenue channel alongside drug commercialization.
NRX-101: Breakthrough Therapy with Expanded Market Potential
Beyond NRX-100, NRXP is advancing NRX-101, an FDA-designated Breakthrough Therapy for suicidal treatment-resistant bipolar depression and chronic pain.
Key attributes:
- Composition-of-matter patent protection globally
- Combines D-cycloserine + low-dose lurasidone
- Designed to mitigate hallucination risk seen with DCS alone
- Newly amended IND to include augmentation of TMS
With projections suggesting 1 million Americans annually may receive TMS by 2030, NRX-101 could capture a significant share of a rapidly expanding neurostimulation market.
NRXP has partnered with Alvogen Pharmaceuticals for development and marketing of NRX-101 in suicidal bipolar depression.
Debt-Free Balance Sheet: Strategic Reset for Growth
More on Colorado Desk
- Artemis Fine Arts and Arte Primitivo's debut collaborative auction was a huge success, totaling nearly $1.5 million
- purelyIV Launches Lab Testing Services in Metro Detroit
- Governor Polis Thanks Executive Director Kim Bimestefer's Service to the State of Colorado and Leadership in Health Care for Coloradans
- Colorado Apprenticeship Month 2026: Spread the Apprenticeship Effect
- On the 296th Anniversary of the Ceremony That Made His Ancestor Emperor, a Cherokee Descendant Publishes the Novel That Restores Him
In December, NRXP eliminated its remaining $5.4 million in balance sheet debt via equity conversion—with no warrants or toxic provisions.
This clean capital structure positions the company for:
- NDA filing preparation
- Clinic expansion
- Commercial scale-up
- Potential strategic partnerships
Analyst Coverage: $34 Price Target
Investment firm D. Boral has issued a Buy rating with a $34 price target, citing regulatory progress, expanded indications, and commercial optionality.
For a company operating at the intersection of FDA reform, real-world evidence utilization, and neuroplastic treatment innovation, this represents a high-conviction thesis on both regulatory and market catalysts.
The Macro Opportunity
According to CDC data, more than 13 million Americans seriously consider suicide each year.
Yet there is no FDA-approved drug indicated specifically for suicidal ideation.
NRXP is positioning itself to:
- Be first-to-label in suicidal depression
- Expand into broader treatment-resistant depression
- Integrate drug + device neuroplastic therapies
- Monetize both pharmaceutical and clinic service channels
This is not a single-asset story—it is a platform strategy targeting one of the largest and most urgent unmet needs in psychiatry.
Investment Thesis Snapshot
- ✅ Fast Track designation for NRX-100
- ✅ 70,000-patient real-world dataset
- ✅ Clear FDA path toward NDA
- ✅ Broader proposed indication
- ✅ Breakthrough Therapy designation (NRX-101)
- ✅ Expanding TMS augmentation indication
- ✅ Strategic partnership with neurocare
- ✅ Debt-free balance sheet
- ✅ Global ketamine market opportunity
For more information on $NRXP visit: https://www.nrxpharma.com/ and https://compasslivemedia.com/case-study/nrx-pharmaceuticals/
Media Contact
Company Name: NRx Pharmaceuticals, Inc. (N A S D A Q: NRXP)
Contact Person: Matthew Duffy, Chief Business Officer
Company Website: https://www.nrxpharma.com/
Email: mduffy@nrxpharma.com
Phone: (484) 254-6134
Home Country: United States
DISCLAIMER: https://corporateads.com/disclaimer/
Disclosure listed on the CorporateAds website
Source: CorporateAds
0 Comments
Latest on Colorado Desk
- Colorado: It's Kickoff Time! Governor Polis Wishes The Denver Summit FC Good Luck In First Home Game
- Homeowner Prep Announces Strategic Language Shift: Replacing "Renters" with "Future Homeowners" to Inspire Wealth-Building Mindsets
- LiposoMore™ Redefines Bioavailability: Joyful Nutritional Launches High-Performance Liposomal Vitamin C and Iron for the Global Supplement Market
- GDE Tree Services Expands Operations into Sydney, NSW
- Tuckwell Machinery Expands CNC Range to Support Australian Cabinet Makers
- The Inner Power of Emotional Self-Leadership
- Dr. Nadene Rose Shares the Secret to True Success: Faith, Obedience, and Divine Purpose
- Colorado: Governor Polis Signs Bills Into Law
- Understanding Unexpected Death: Why Independent Autopsies Matter in Cases Without Clear Cause
- Governor Jared Polis Hands Out 3 Millionth Book with Colorado Department of Early Childhood Highlighting the Imagination Library of Colorado's Statewide Impact
- Epic Pictures Group Sets North American Release Date for the Thriller NO ORDINARY HEIST
- Boulder SEO Marketing Opens Louisville Satellite Office and Announces Local SEO Training with AI SEO Expert Chris Raulf
- Colorado: Governor Polis Celebrates Overall Population Increase, Population Increased in Counties with Most Housing Construction
- Award-Winning REALTOR® Paige Coker Joins Corcoran DeRonja Real Estate
- Denver's Largest Neurodiversity Fair Names National Medical Supply as Visionary Sponsor
- Over 98% of crypto owners globally don't declare taxes, new report find
- TicTac Group acquires French EdTech company Distrisoft
- Colorado: What Local, Community, and Housing Leaders Are Saying About Governor Polis and the Legislature Taking Action to Build More Housing
- Colorado: Governor Polis Signs Laws Breaking Down Barriers to New Homes Coloradans Can Afford
- Mark Dobosz Makes Donorassess.org Free To Every Nonprofit On The Planet