Trending...
- NoviSign Sponsoring VARTECH 2025 - the B2B IT channel's #1 event - 108
- Athena Forge (ATFG) Introduces Advanced Token for Technology-Driven Financial Ecosystem - 107
- Green Office Partner Named #1 Best Place to Work in Chicago by Crain's for 2025 - 104
MUNICH - ColoradoDesk -- OncoBeta® GmbH, a medical device company specialising in innovative epidermal radioisotope therapies, has announced 12-months interim results from its international Phase IV, multi-centre clinical trial evaluating the efficacy and safety of Rhenium-SCT® (Skin Cancer Therapy) in patients with non-melanoma skin cancer (NMSC).
The EPIC-Skin Study (Efficacy of Personalised Irradiation with Rhenium-SCT for the Treatment of Non-Melanoma Skin Cancer) is designed to assess treatment efficacy and safety, as well as key patient-reported outcomes including quality of life, treatment comfort, and cosmetic results.1 The study is based on the clinically validated effect of the beta-emitter rhenium-188 in treating basal cell carcinoma (BCC) and squamous cell carcinoma (SCC).2
Treatment data is available for 184 adult patients (with 254 lesions) across 7 study centres in Australia, Austria, Germany, the United Kingdom, and South Africa. All participants had histologically confirmed stage I or II NMSC. The median patient age was 70.3 years (range 27–95 years), and 53.3% of participants were male. Patients had one (71.2%), two (19.6%), or three (9.2%) lesions treated, which were located on the head & neck (11.8%), trunk (11.8%), lower limbs (11.8%), or upper limbs (9.1%).1
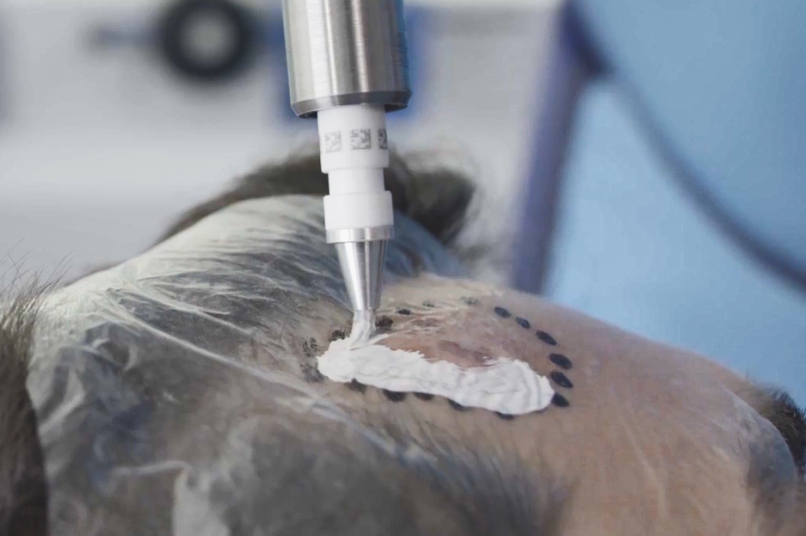
Treatment & Methodology
Rhenium-SCT was administered as a single 50-Gy topical dose of rhenium-188 embedded in a resin applied via adhesive film to the lesion site. Efficacy was assessed using modified RECIST criteria, at 12 months follow-up. Quality of life was assessed using the Skin Cancer Index (SCI) at baseline, 6 months, and 12 months. Treatment comfort was assessed using a patient questionnaire, whilst patient- and clinician-evaluated cosmetic outcomes were determined using a visual analogue scale (VAS) grading of 0-10 (0 = very poor; 10 = no wound detectable). Safety was assessed via treatment-emergent adverse events (TEAEs) and CTCAE (Common Terminology Criteria for Adverse Events) grading.
Key 12-Month Results:1
These findings confirm that Rhenium-SCT®, administered in a single session, delivers sustained high efficacy and improved quality of life, with excellent cosmetic outcomes and minimal adverse effects.
More on Colorado Desk
"Rhenium-SCT® has consistently demonstrated effectiveness and safety in prior studies," said Dr. Gerhard Dahlhoff, Medical Director at OncoBeta®. "This 12-month interim data further reinforces its value as a non-invasive, targeted treatment option for NMSC, particularly for patients seeking alternatives to surgery due to cosmetic or health-related concerns."
"The 12-month results from the EPIC-Skin Study represent a major milestone for OncoBeta®," added Shannon D. Brown III, CCO OncoBeta & Managing Director Europe. "These results support the efficacy, safety, and patient satisfaction associated with Rhenium-SCT®. These are the three foundational pillars of our patient-first vision, emphasising not only clinical outcomes, but also the patient experience in achieving them."
_
About the Rhenium-SCT® (Skin Cancer Therapy)
Non-melanoma skin cancer (NMSC) is the most common form of cancer in humans.3 The most common cause of NMSC is sun exposure, while other predisposing factors include genetic skin conditions and immunosuppressive diseases or treatments.4
The Rhenium-SCT is a painless*, non-invasive‡ therapy that provides aesthetic results, even in cases otherwise considered difficult to treat.1,5,6 The Rhenium-SCT utilises the radioisotope Rhenium-188 in an epidermal application with optimal properties for the treatment of NMSCs (non-melanoma skin cancers). The Rhenium-SCT is a precise, personalised therapy2 that is only applied to the area needed to treat without affecting the healthy tissue. The specially designed device ensures the Rhenium-SCT compound never comes in direct contact with the patient's skin and the application is safe and simple for the applying physician. Scar-free healing of the treated lesion area and the regeneration of healthy tissue occurs usually within a few weeks after treatment.6-8
About OncoBeta®
OncoBeta® with its headquarters located in Garching near Munich, Germany, is a privately held medical device company, specializing in the development and commercialization of state-of-the-art, innovative therapies. Since its foundation, OncoBeta® has concentrated its efforts on the development, regulatory approval(s) and commercialization of the epidermal radioisotope therapy Rhenium-SCT® (Skin Cancer Therapy), targeting non-melanoma skin cancers and keloid scars. OncoBeta® has perfected the customized application and device management system in conformity with all health, safety, and environmental protection regulatory standards.
More on Colorado Desk
Find out more about the Rhenium-SCT® at www.oncobeta.com
Follow us on social media:
LinkedIn: https://www.linkedin.com/company/oncobeta-gmbh/
Facebook: https://www.facebook.com/oncobeta/
Instagram: https://www.instagram.com/oncobeta_gmbh/
Forward-looking statements
This announcement includes forward-looking statements that involve risks, uncertainties, and other factors, many of which are outside of OncoBeta's control, and which could cause actual results to differ materially from the results discussed in the forward-looking statements. Forward-looking statements include statements concerning OncoBeta's plans, objectives, goals, future events, performance and/or other information that is not historical information. All such forward-looking statements are expressly qualified by these cautionary statements and any other cautionary statements which may accompany the forward-looking statements. OncoBeta® undertakes no obligation to publicly update or revise forward-looking statements to reflect subsequent events or circumstances after the date made, except as required by law.
*No pain reported during procedure.6
‡No direct skin contact or incisions.6
References
The EPIC-Skin Study (Efficacy of Personalised Irradiation with Rhenium-SCT for the Treatment of Non-Melanoma Skin Cancer) is designed to assess treatment efficacy and safety, as well as key patient-reported outcomes including quality of life, treatment comfort, and cosmetic results.1 The study is based on the clinically validated effect of the beta-emitter rhenium-188 in treating basal cell carcinoma (BCC) and squamous cell carcinoma (SCC).2
Treatment data is available for 184 adult patients (with 254 lesions) across 7 study centres in Australia, Austria, Germany, the United Kingdom, and South Africa. All participants had histologically confirmed stage I or II NMSC. The median patient age was 70.3 years (range 27–95 years), and 53.3% of participants were male. Patients had one (71.2%), two (19.6%), or three (9.2%) lesions treated, which were located on the head & neck (11.8%), trunk (11.8%), lower limbs (11.8%), or upper limbs (9.1%).1
Treatment & Methodology
Rhenium-SCT was administered as a single 50-Gy topical dose of rhenium-188 embedded in a resin applied via adhesive film to the lesion site. Efficacy was assessed using modified RECIST criteria, at 12 months follow-up. Quality of life was assessed using the Skin Cancer Index (SCI) at baseline, 6 months, and 12 months. Treatment comfort was assessed using a patient questionnaire, whilst patient- and clinician-evaluated cosmetic outcomes were determined using a visual analogue scale (VAS) grading of 0-10 (0 = very poor; 10 = no wound detectable). Safety was assessed via treatment-emergent adverse events (TEAEs) and CTCAE (Common Terminology Criteria for Adverse Events) grading.
Key 12-Month Results:1
- Overall response rate: 97.3%
- Complete response rate: 94.1%
- Partial response rate: 3.2%
- Mean improvement in quality of life: +10.55 points from baseline
- Pain/discomfort during treatment: None reported
- Cosmetic outcomes: Favourable results reported by both patients and clinicians
- Most common toxicity (12-month): Grade 1 hypopigmentation (60.4%)
- No CTCAE toxicities above Grade 2 observed at 12 months
These findings confirm that Rhenium-SCT®, administered in a single session, delivers sustained high efficacy and improved quality of life, with excellent cosmetic outcomes and minimal adverse effects.
More on Colorado Desk
- Colorado: Host Your Holiday Celebration at The Governor's Residence at Boettcher Mansion
- Denver LASIK & Vision Correction Leader 20/20 Institute Sets New Standard for Concierge-Level Eye Surgery in Colorado
- New Leadership and Renovations Usher in Next Chapter for Sunrise Manor
- Who Will Win the 2025 WNBA Finals? OddsTrader Shares Live Betting Odds and Projections
- Geeks5g Creative Marketing: The Powerhouse Behind Business Growth
"Rhenium-SCT® has consistently demonstrated effectiveness and safety in prior studies," said Dr. Gerhard Dahlhoff, Medical Director at OncoBeta®. "This 12-month interim data further reinforces its value as a non-invasive, targeted treatment option for NMSC, particularly for patients seeking alternatives to surgery due to cosmetic or health-related concerns."
"The 12-month results from the EPIC-Skin Study represent a major milestone for OncoBeta®," added Shannon D. Brown III, CCO OncoBeta & Managing Director Europe. "These results support the efficacy, safety, and patient satisfaction associated with Rhenium-SCT®. These are the three foundational pillars of our patient-first vision, emphasising not only clinical outcomes, but also the patient experience in achieving them."
_
About the Rhenium-SCT® (Skin Cancer Therapy)
Non-melanoma skin cancer (NMSC) is the most common form of cancer in humans.3 The most common cause of NMSC is sun exposure, while other predisposing factors include genetic skin conditions and immunosuppressive diseases or treatments.4
The Rhenium-SCT is a painless*, non-invasive‡ therapy that provides aesthetic results, even in cases otherwise considered difficult to treat.1,5,6 The Rhenium-SCT utilises the radioisotope Rhenium-188 in an epidermal application with optimal properties for the treatment of NMSCs (non-melanoma skin cancers). The Rhenium-SCT is a precise, personalised therapy2 that is only applied to the area needed to treat without affecting the healthy tissue. The specially designed device ensures the Rhenium-SCT compound never comes in direct contact with the patient's skin and the application is safe and simple for the applying physician. Scar-free healing of the treated lesion area and the regeneration of healthy tissue occurs usually within a few weeks after treatment.6-8
About OncoBeta®
OncoBeta® with its headquarters located in Garching near Munich, Germany, is a privately held medical device company, specializing in the development and commercialization of state-of-the-art, innovative therapies. Since its foundation, OncoBeta® has concentrated its efforts on the development, regulatory approval(s) and commercialization of the epidermal radioisotope therapy Rhenium-SCT® (Skin Cancer Therapy), targeting non-melanoma skin cancers and keloid scars. OncoBeta® has perfected the customized application and device management system in conformity with all health, safety, and environmental protection regulatory standards.
More on Colorado Desk
- Governor Polis and Colorado State Demography Office Release Analysis on the Current State of Colorado's Housing Shortfall
- Colorado: Lt. Governor Dianne Primavera Honored with National Award for Leadership in Service
- Agemin Unveils Breakthrough AI Model for Biometric Age Estimation, Setting New Standards in Online Child Safety
- InventHelp Inventor Develops Portable Hybrid Grow System (DNV-640)
- Strategic Partnerships with Defiant Space Corp and Emtel Energy USA Powerfully Enhance Solar Tech Leader with NASA Agreements: Ascent Solar $ASTI
Find out more about the Rhenium-SCT® at www.oncobeta.com
Follow us on social media:
LinkedIn: https://www.linkedin.com/company/oncobeta-gmbh/
Facebook: https://www.facebook.com/oncobeta/
Instagram: https://www.instagram.com/oncobeta_gmbh/
Forward-looking statements
This announcement includes forward-looking statements that involve risks, uncertainties, and other factors, many of which are outside of OncoBeta's control, and which could cause actual results to differ materially from the results discussed in the forward-looking statements. Forward-looking statements include statements concerning OncoBeta's plans, objectives, goals, future events, performance and/or other information that is not historical information. All such forward-looking statements are expressly qualified by these cautionary statements and any other cautionary statements which may accompany the forward-looking statements. OncoBeta® undertakes no obligation to publicly update or revise forward-looking statements to reflect subsequent events or circumstances after the date made, except as required by law.
*No pain reported during procedure.6
‡No direct skin contact or incisions.6
References
- Cardaci G, et al. Adv Radiat Oncol. 2025;10(7):101802.
- Cipriani C, et al. J Dermatol Treat. 2022;33(2):969–975. Epub 22 Jul 2020.
- Ciążyńska M, et al. Sci Rep. 2021;11(1):4337.
- Cancer.net. Skin Cancer (Non-Melanoma): Risk Factors and Prevention. February 2022. https://www.cancer.net/cancer-types/skin-cancer-non-melanoma/risk-factors-and-prevention (accessed June 2025).
- Tietze JK, et al. Clin Nuc Med. 2023;48(10):869–876.
- Castellucci P, et al. Eur J Nucl Med Mol Imaging. 2021;48(5):1511–1521.
- Cipriani C, et al. Int J Nucl Med. 2017;114–112.
- Sedda AF, et al. Clin Exp Dermatol. 2008;33(6):745–749.
Source: OncoBeta GmbH
Filed Under: Health
0 Comments
Latest on Colorado Desk
- Tour Napa Like a Local: Vines of Napa Valley Wine Passport AKA Vine Pass Unlocks Hidden Gems
- Dental Surgical Center Accepts Sedation Patients with Medicaid for MD, WV, PA and DC
- Backed by Local Launches FREE City Hubs to Empower Small Businesses in Colorado, Washington, & Oreg
- Colorado: Governor Polis Joins Governors' Call to Congress to Extend Health Care Tax Credits and Lower Costs for Families
- Colorado Leading the Way: New Study Finds Land Use Reforms, Many of Which Colorado Has Already Taken, Popular Especially Among Seniors
- Colorado: Governor Polis Releases Statement on Passing of American Icon and Actor Robert Redford
- Sloan's Lake Dental Launches New Website to Enhance Patient Experience and Access to Modern Dental Care
- Only 3 Weeks Left till the Start of the OpenSSL Conference 2025
- ENTOUCH Completes $50 million Funding Round
- Teaming Agreement with Emtel Energy USA to Advance Thin-Film PV Energy Storage Capabilities; NASA agreements for Solar Space Tech; Ascent Solar $ASTI
- Nebuvex Acknowledges Platform "Too Secure" for Anonymous Traders; Institutional Investors Disagree
- From Tokyo to Berlin: FreeTo.Chat Unites Cultures with the World's First Confession VRX — EmojiStream™
- AZETHIO Launches Multi-Million Dollar User Protection Initiative Following Unprecedented Platform Growth
- Matecrypt Observes South American Cryptocurrency Adoption Surge Amid Economic Shifts
- Assent Uncovers Over 695 Unique PFAS Across Global Supply Chains as Regulations Increase
- Cryptocurrency Quarterly Trading Volume Surpasses $15 Trillion Record High as BrazilNex Acknowledges Industry 'Growing Pains' Amid Market Speculation
- AHRFD Initiates Legal Proceedings Against Anwalt.de for Publishing Defamatory and False Content
- New Analysis Reveals the Complex Forces Driving the 'Great Human Reshuffle'
- Stillwater Architecture Offers Principal-Led Custom Home Services in Boulder & Big Sky
- NetDiligence® Adds ZeroDown® SafeHouse for WordPress™ to eRiskHub® Platform