Trending...
- Danholm Collection Launches Boutique Luxury Real Estate Brokerage in Central Florida
- Claude Riveloux Review 2026: How the $10B Fund Manager Dispels 'Scam' Rumors Through Education
- Food Journal Magazine Raises the Standard for Restaurant Reviews in Los Angeles
FAYETTEVILLE, Ark. - ColoradoDesk -- Lineus Medical is now officially registered in the United Kingdom, enabling the company to begin distributing SafeBreak® Vascular within the UK healthcare market. UK registration represents an important step in Lineus Medical's international expansion strategy, further extending access to SafeBreak Vascular for hospitals and clinicians on a global scale. With regulatory requirements in place, Lineus Medical can now work with distribution partners to bring its breakaway IV technology to the UK.
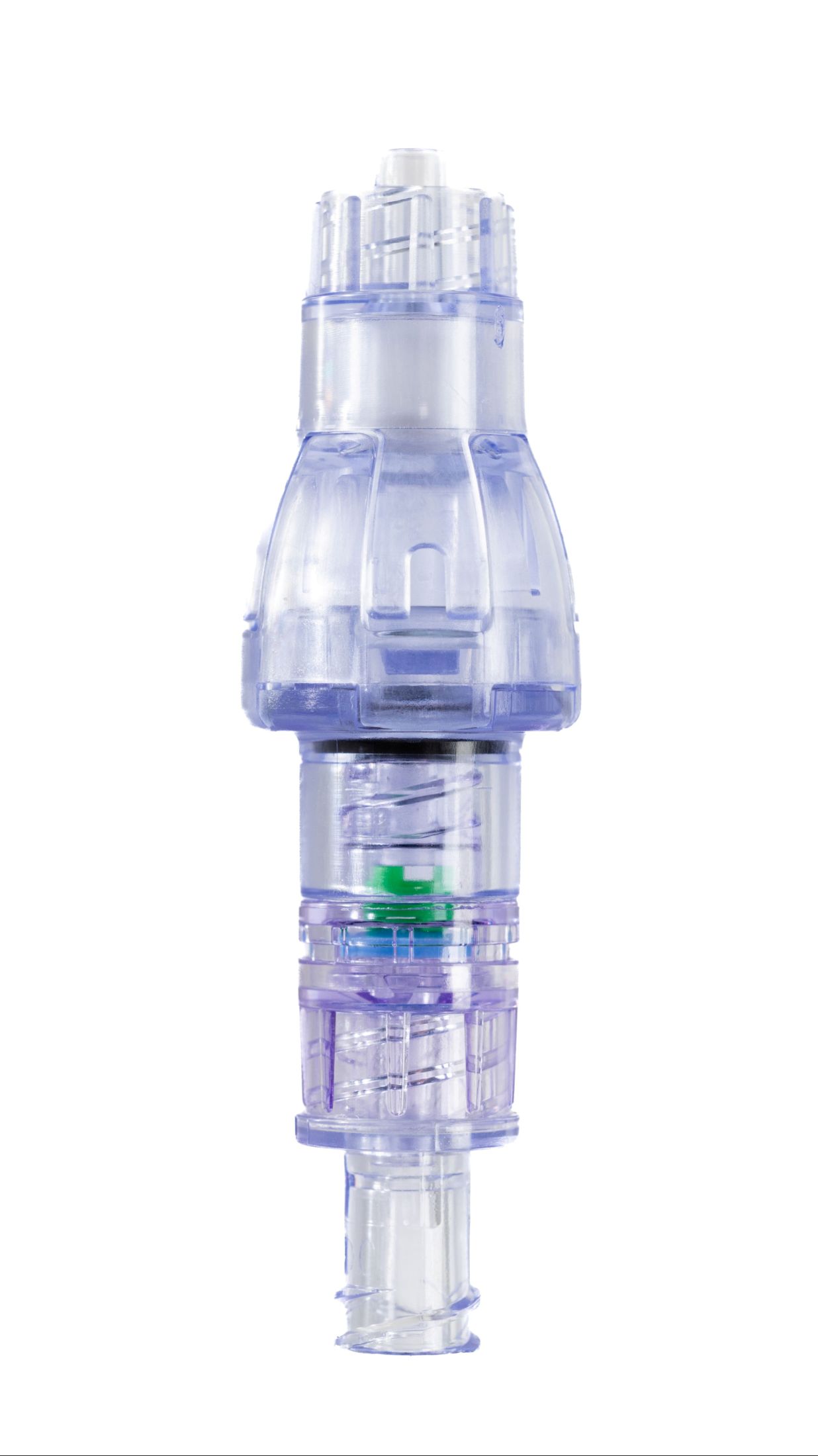
SafeBreak Vascular is a breakaway device for IV lines clinically proven to reduce IV complications.¹ When a harmful force is placed on the line, SafeBreak intentionally separates to remove the damaging force and protect the patient's IV. When separation occurs, valves on both ends of the device close to prevent medication spills from the pump and blood loss from the patient. Patients avoid additional needlesticks, nurses save time, and hospitals save money.¹
More on Colorado Desk
"Completing UK registration is another meaningful milestone as we continue to expand access to SafeBreak globally," said Vance Clement, CEO of Lineus Medical. "Each new market brings us closer to our mission of removing the pains associated with IV lines and improving patient safety across healthcare systems worldwide. UK registration allows SafeBreak to reach another 69 million people."
"This registration allows us to move forward with distribution planning in the UK and supports our broader international commercialization efforts," said Larry Hayes, Chief Commercial Officer of Lineus Medical. "We are focused on working with the right partners to ensure SafeBreak Vascular is accessible to clinicians who are looking to reduce IV complications and improve care at the bedside."
About Lineus Medical:
Lineus Medical is the developer of SafeBreak® Vascular, a breakaway technology proven to reduce IV restarts and IV complications.1 Our mission is to remove the pains associated with medical lines. More information about Lineus Medical can be found at www.lineusmed.com. Follow Lineus Medical on LinkedIn, Facebook, and Instagram.
References
SafeBreak Vascular is a breakaway device for IV lines clinically proven to reduce IV complications.¹ When a harmful force is placed on the line, SafeBreak intentionally separates to remove the damaging force and protect the patient's IV. When separation occurs, valves on both ends of the device close to prevent medication spills from the pump and blood loss from the patient. Patients avoid additional needlesticks, nurses save time, and hospitals save money.¹
More on Colorado Desk
- ASI Hosts 2026 Executive Business Summit for Global Partner Community
- Pastor Saeed Abedini Releases THE TRUTH – Volume 1, A Deeply Personal Story of Faith, Struggle, and Redemption
- New Book Warring From the Standpoint of the Throne Room Calls Believers to Pray From Victory
- Boulder SEO Marketing Cracks the SEO Code for Apartment Companies and Multifamily Operators Nationwide
- Scotch Whisky Market Dislocation Creates Compelling Entry Opportunity for Long-Term Investors
"Completing UK registration is another meaningful milestone as we continue to expand access to SafeBreak globally," said Vance Clement, CEO of Lineus Medical. "Each new market brings us closer to our mission of removing the pains associated with IV lines and improving patient safety across healthcare systems worldwide. UK registration allows SafeBreak to reach another 69 million people."
"This registration allows us to move forward with distribution planning in the UK and supports our broader international commercialization efforts," said Larry Hayes, Chief Commercial Officer of Lineus Medical. "We are focused on working with the right partners to ensure SafeBreak Vascular is accessible to clinicians who are looking to reduce IV complications and improve care at the bedside."
About Lineus Medical:
Lineus Medical is the developer of SafeBreak® Vascular, a breakaway technology proven to reduce IV restarts and IV complications.1 Our mission is to remove the pains associated with medical lines. More information about Lineus Medical can be found at www.lineusmed.com. Follow Lineus Medical on LinkedIn, Facebook, and Instagram.
References
- Data on file.
Source: Lineus Medical
Filed Under: Medical, Technology
0 Comments
Latest on Colorado Desk
- Colorado: Governor Polis, Fellow Governors Call on Congress to Reject Costly, Unlawful Tariffs
- Phase One Landscapes Launches Updated Denver Patios and Decks Page
- $6 Million Funding Secured as Retail Expansion, Operational Streamlining, and Asset-Light Strategy Position the Company for Accelerated Growth $SOWG
- Sleep Basil Launches Revamped Memory Foam Mattress Collection Page, Elevating the Mattress Shopping Experience for Denver and Beyond
- The "Unsexy" Business Quietly Creating 130+ New Entrepreneurs Across America — From Alaska to Puerto Rico
- Veteran Launches GTG Energy: Nicotine-Free Pouch as Americans Rethink Addiction, Focus, and What Fuels Performance
- Colorado: Lt. Governor Dianne Primavera Named 2026 HIMSS Changemaker in Health Award Recipient
- RecallSentry™ App Launch — Your Home Safety Hub — Free on iOS & Android
- Award-Winning Director Crystal J. Huang's Under-$50K Film "The Ritual House" Wins Best Horror Feature at Golden State Film Festival
- Grads aren't getting hired — here's what we're doing about it
- Colorado Springs: Garden of the Gods Road reduced to one lane for overnight work Wednesday
- Colorado Springs: Statement from Councilman Tom Bailey Regarding Results of Recall Petition
- Colorado Springs: City continues investment in North Cheyenne Cañon with Starsmore Visitor and Nature Center improvements
- Colorado Springs: City Clerk declares recall petition for District 2 Councilmember sufficient
- K2 Integrity Enhances Technology Capabilities Through Acquisition of Leviathan Security Group
- #WeAreGreekWarriors Comes to Detroit in Celebration of Women's History Month
- Colorado Tourism Office Unveils 2026 Tourism Management Grant Awardees
- Energywise Solutions and Pickleball Pros Partner to Bring More Energy and Visibility to Pickleball Clubs
- Buildout Launches CRM, Completing the Industry's First AI-Powered End-to-End Deal Engine for CRE
- The Franchise King® Releases Free Guide for Nervous Buyers